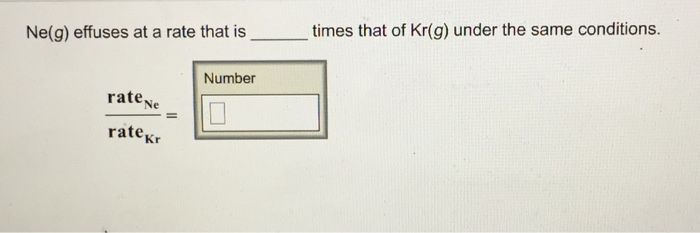
Ne(g) Effuses At A Rate That Is ______ Times That Of Kr(g) Under The Same Conditions.
Khalid Young Dumb And Broke, Khalid - Young Dumb & Broke (Official Video), 5.68 MB, 04:08, 860,329,150, KhalidVEVO, 2017-08-01T14:00:02.000000Z, 19, Khalid – Young Dumb & Broke Lyrics | Genius Lyrics, genius.com, 1000 x 1000, jpeg, dumb broke young khalid lyrics genius, 20, khalid-young-dumb-and-broke, Kampion
Ne(g) effuses at a rate that is times that of kr(g) under the same conditions. Ne(g) effuses at a rate that is times that of kr(g) under the same conditions. Rate co/rate kr= (83. 7 amu/28. 02 amu)^1/2 = 1. 73 times faster than kr this is found using graham’s law of effusion. Basically to find the ratio of co’s rate of effusion to kr’s.
And for neon it is 20. 18 g per mole. That comes out to be under road 6. 51 which is approximately equal to 2. 55. Solution for times that of kr(g) under the same conditions. Ne(g) effuses at a rate that is ratee rater. Skip to main content. First week only $4. 99! N2(g) effuses at a rate that is _____ times that of kr(g) under the same conditions. 1. 1. 59, 2. 1. 43, 3. 1. 73, 4. 2. 16 Solution for ne(g) effuses at a rate that is _____ times that of kr(g) under the same conditions.
Solved: Ne(g) Effuses At A Rate That Is Times That Of Kr(g... | Chegg.com

PPT - Gases PowerPoint Presentation, free download - ID:2215121

PPT - Gases PowerPoint Presentation, free download - ID:2215121

PPT - Gases PowerPoint Presentation, free download - ID:2215121
