A Water Solution Of Sodium Acetate Is Basic Because...

Khalid Young Dumb And Broke, Khalid - Young Dumb & Broke (Official Video), 5.68 MB, 04:08, 860,329,150, KhalidVEVO, 2017-08-01T14:00:02.000000Z, 19, Khalid – Young Dumb & Broke Lyrics | Genius Lyrics, genius.com, 1000 x 1000, jpeg, dumb broke young khalid lyrics genius, 20, khalid-young-dumb-and-broke, Kampion
Solution verified by toppr in aqueous solution sodium acetate ( c h 3 c o o n a ) dissociates as under and forms weak acid ( c h 3 c o o h ) and strong base ( n a o h ) It is a sodium salt of acetic acid or sodium acetate anhydrous (i. e. , lacking water of hydration) or sodium ethanoate. It is easily soluble in water and alcohol and is hygroscopic in nature. The equation clearly states it is basic ( slightly ) , but the equation does not look quite complete.
The epa has set limits on how much sodium acetate can be added to the water supply. Acetic acid reacts with sodium carbonate or sodium hydroxide to form a basic solution of sodium acetate which acts as buffer with acetic acid. B) acidic, because the acetate ion is acidic. It is widely used across a number of industrial sectors. It is hygroscopic in nature and easily. The aqueous solution of sodium acetate is basic. Share it on facebook twitter email. 1 answer +1 vote. Answered jul 3, 2020 by amankumar.
A water solution of sodium acetate is basic because 1 the conjugate

A water solution of sodium acetate is basic because...... | Clutch Prep

Why does the solution of sodium acetate give more concentration of
Solved: For The Following Compound (sodium Acetate), Show | Chegg.com

PPT - Relationship Between K a and K b PowerPoint Presentation, free

Solved: 3. One Way To Drive A Fisher Esterification Reacti... | Chegg.com
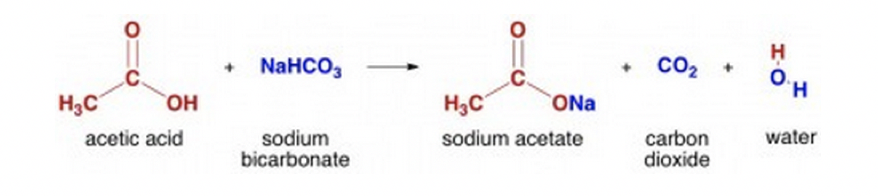
15.9 Hydrolysis of Esters | The Basics of General, Organic, and

Activity 2.14 Collect the following salt samples - sodium chloride

15.8: Hydrolysis of Esters - Chemistry LibreTexts
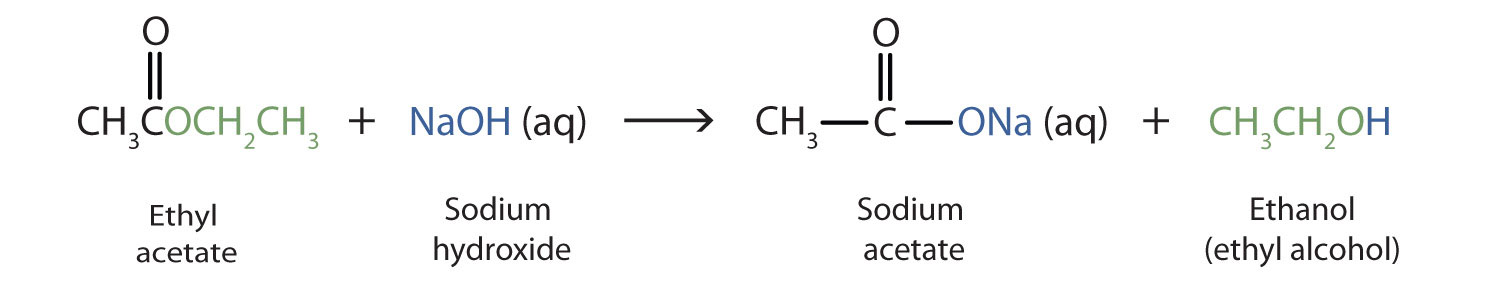
PPT - Acid/Base Properties of Salts PowerPoint Presentation, free
